
In current practice, whether an act of administrative approval can be accommodated under Bolar exemption, has not been generally agreed upon among National Medical Products Administration (NMPA), courts, brand-name pharmaceuticals, and me-too drug manufacturers. In this haze there emerges uncertainties and potential chaos in the patent drug market.
2019 is a tough year for brand-name pharmaceuticals. From January till October, 10 me-too drugs have been approved for marketing within the patent terms of the relevant brand-name drug compounds, almost by a surprising rate of "one fast follow-on per month" (see Fig. 1). An insider explains to China IP that once approved for marketing by the NMPA, it means the drug company can market its products, e.g., to be qualified for participation in government procurement for 4+7 cities and the online bidding procurement. China IP reporters learned in interviews that a few me-too drug manufacturers in various provinces that have received approvals have successfully been listed in the procurement programs or have won biddings.
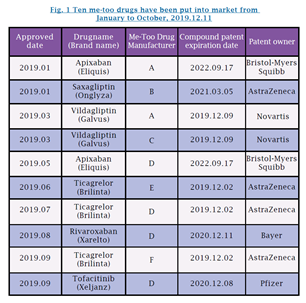
Under Management Measure for Drug Registration (MMDR), for Chinese patented drugs, other applicants may file applications for registration within 2 years prior to the patent expiration. The NMPA conducts examination by the Measure, and gives approval for production or importation upon patent expiration. The 2008 third revision of the Patent Law introduced the Bolar Exemption, which is a collision product of US drug administration system and patent system, specifying that "It shall not be an act of patent infringement to exploit a patent for purposes of conducting clinic experimentation on drugs or medical appliances, and for applying for registration." A patent administrator from a drug company said, "The Bolar Exemption has turned the MMDR's 2 years requirement a lip service only, which is long abandoned by the MNPA. Under the circumstances that me-too drug manufacturers take theirs own risks, it seems that there is no legal barrier for marketing a me-too drug before the patent expires."
Regrettably, all interviewed patent administrators wish to remain anonymous because of the sensitive situation. But the common concern reflected in the interviews is that due to lack of timely and effective legal remedies, there is nothing brand-name pharmaceuticals could do in the process of prior approval of me-too drugs except watching me-too drug manufacturers go through the process and start marketing. Of course, there are a few me-too drug manufacturers that are cautious for large scale marketing activities even though they have received approval.
The unique event of "one fast follow-on per month" of 2019, may, by insiders' estimate, be related to the government's effort to push for accessibility for drugs on the one hand, and on the other, to the speed-up of NMPA's examinations and approvals. But what is perplexing is that, since the Opinions on Deepening the Evaluation and Approval Systems and Encouraging Innovation on Drugs and Medical Devices 2017 jointly issued by The General Office of the CPC Central Committee and the General Office of the State Council, the pharmaceutical industry has been probing to set up a series of measures for reform, including establishment of patent linkage system for drugs, conducting pilot tests for compensating drug patent terms, as well as improvement and implementation of protection scheme for drug testing data, etc. "Policy having been well oriented, as though more emphasis would be placed on IP, yet reality is practically more distant from it," said Li Ming (pseudonym), general patent supervisor of a foreign pharmaceutical enterprise who wished to remain anonymous, "the current situation make people worry and even fear that it will become a trend in the future. There are no clear rules for the entire industry. People work in confusion, losing the direction in the haze."
Cheng Yongshun, director of Beijing Intellectual Property Institute and former deputy director of the Intellectual Property Division of Beijing High People's Court, has in recent years been actively pushing for implementation of a patent linkage system. He told China IP, "administrative approval of me-too drugs does not preclude antitrust violation for marketing within patent terms. In case of an infringement, it will be the patent owners, me-too drug manufacturers and consumers (patients) that will foot the bill together for the damage. The consumer, after buying cheap me-too drugs, will have to turn to the pricy brand-name drugs again because of the infringement problem, which is not only bad for patients, but also put pressure on the judiciary for determining infringement."
A reading of the Bolar exemption: should administrative approval fall under the Bolar exemption?
Li Ming told China IP that prior to the other side obtains approval for me-too drugs, his company had communicated with NMPA by letters for many times, reasoning that its brand-name drug is under effective compound patent, and there are "quasi-patent linkage" laws that no me-too drugs can be applied for registration and no approval of me-too drugs may be granted within 2 years before expiration of patent under MMDR. "But either our letters went unanswered, or we got occasional response, saying that the administrative activities of advance approval for marketing falls under the Bolar Exemption.
As to the 2-year provision in approving drug marketing involving patent problems, an industry insider told China IP that despite the MMDR regulation, it has long been discontinued in practice. The IP general supervisor of AstraZeneca China and Asia said that some me-too drugs start application for clinical bioequivalence test even when the brand-name drug is still in clinical test phase, to be yet approved in China. China IP found during interviews the patent status of the major brand-name drugs to be followed on:
The administrative litigation of patent invalidity of AstraZeneca's Ticagrelor was reversed by Beijing High People's Court in the second instance in 2018, which vacated the lower court's decision of invalidity. This case was listed by the Supreme People's Court as one of the 50 Typical IP Cases of 2018.
The Vildagliptin compound patent of Novartis was upheld by the Patent Reexamination Board after multiple patent invalidity challenges; and Rivaroxaban (Xarelto), Bayer's compound patent has never before been challenged. "Regrettably, this cannot even stop the me-too drugs from getting the advance approval for marketing."
"In fact," Liu Hongqiang, Bayer's IP administrator for Greater China said, "the advance approval also puts me-too drug makers in an awkward position. For me-too makers, the real competitor is not the brand-name drug maker, but other immediate follow-on drug makers. When a me-too drug maker obtains an advance approval before the original brand-name drug's patent expires, the competition pressure and market enticement may very likely prompt these early me-too drugs to take the risk of go on market or bring forth a challenge of the patent for no obvious reason." What is worth noting is that, as a heavyweight market hit from Bayer, the Xarelto patent has never before been challenged, but it now faces invalidity challenges at a time when the first me-too drug obtains an advance approval and the patent expiration is less than one year.
Some think that in order to resolve the drug accessibility issue, NMPA may, prior to expiration of drug patent, approve marketing application for me-too drugs. "But in reality," says Li Ming, "the sale of brand-name drugs is a process of gradual increase, and more often than not, the last few years toward patent expiration may be the vital moment for profit. One year, or even just a few months, would have tremendous commercial impact and influence on us. This is common in the industry."
Could the introduce of Bolar exemption into Patent Law invalidate the 2 year requirement of MMDR? The reporter found that there had been no official reading of the Bolar exemption to be applied on the administrative conduct of approving drug marketing by the NMPA.
The third revision of the Patent Law of 2008 modified Article 69 as: "The following shall not be deemed as patent infringement: … (5) Any person produces, uses, or imports patented drugs or patented medical apparatus and instruments, for the purpose of providing information required for administrative examination and approval, or any other person imports or produces patented drugs or patented medical apparatus and instruments especially for that person." This so-called Chinese version of "Bolar exemption" is to resolve the tension caused by the coexistence of the patent system and the system for supervising and managing medicinal products and medical appliances. As is explained, Bolar exemption originated from American patent law, and as an exemption to patent infringement, it exonerates me-too drug manufacturers from infringement liabilities for conducting clinical tests and collecting data for approval before patent expiration. An industry insider explained that, because the drug approval takes a long time, the marketing of a me-too drug would be long after the patent expires if it has to wait until the patent expires before starting the testing and approval process, which in effect prolongs the patent protection of the drug. Bolar Exemption resolves this problem so that the cheaper me-too drugs can be marketed in time when the brand-name drugs patents expire.
But in current practice, whether the act of administrative approval falls under the Bolar Exemption is a question to which the NMPA, courts, brand-name drug pharmaceuticals and me-too drug manufacturers could not reach an unanimous agreement, which leads to the market instability and potential chaos for drug patents.
Prof. Zhang Guangliang of Remin Law School and former official in charge of IP division of Beijing No.1 Intermediate People's Court, indicated that the Bolar Exemption is totally inconsonant with the legislative intent and the current provisions of the law. The original legislative intent of the Bolar Exemption was for the mere purpose of approving a drug for marketing, and may not be extended to marketing activity itself. Cheng Yongshun also indicated that "precisely because the Bolar Exemption is an exception to patent infringement, it should be appliedmuch narrowly to me-too drug manufacturers rather than drug authorities; what the Bolar Exemption targets ought to be research activities of me-too drug manufacturers before applying for approval, rather than production activities or official approval activities. Therefore the prior approval activities of NMPA should not fall under Bolar Exemption."
The risk of me-too drugs sold on market: offer for sale may constitute patent infringement
On November 15, 2018, the state initiated concentrated procurement pilot programs covering 11 cities including Beijing, Tianjin, Shanghai, Chongqing, Shenyang and Dalian (4+7 cities). The piloted areas appointed representatives to form a joint procurement office, representing the public medical organizations for concentrated procurement. The first round of 4+7 cities' procurement made up 25%~30% of national sales. According to the 4+7 Cities Concentrated Drug Procurement Document, drugs required for approval refer to the listed drugs that have obtained valid domestic registration documents within the scope of the procurement catalogue. "Relative documents do not mention anything about patent as a condition for application qualification, but rather place emphasis on whether a drug manufacturer has obtained effective approval for the drugs specified by the procurement scheme." Yan Jianzhou, a project researcher of the research center for national drug policy and ecosystem of China Pharmaceutical University said, "once a me-too drug is approved for marketing, even if before original brand-name drug's patent expires, it will not affect the marketing qualification of relevant products, and manufacturers can smoothly place the products into the market, and participate in the procurement for 4+7 cities, online bidding and other marketing activities."
During the interviews, Xu Feng and Li Ming said that quite a few approved me-too drugs have been listed in various provincial procurement catalogues, or have won online bidding procurement. "This is not just a concern, but a done deal."
Cheng Yongshun believes whether the quantitative procurement for 4+7 cities or the online bidding procurement are in essence offer for sale activities prohibited by Article 11(1) of the Patent Law as patent infringement. Offer for sale, under Art. 24 of Several Provisions of the Supreme People's Court on Issues concerning the Application of Law in the Trial of Cases on Patent Disputes (2015 Amendment), refers to the expression for sale of products by advertisement, display in shop windows, or in exhibition on trade fairs. "Apart from the activities clearly indicated in the judicial interpretations, offer for sale may also be understood as certain activities conducted prior to making actual sales, including advertisement, exhibition, public display, shopping catalogue, bidding announcement, reaching sale agreement and other activities that indicate sale of products." Cheng further explained, "once quantitative procurement for 4+7 cities or online bidding procurement is confirmed, an agreement is reached with the government, and the underlying products may officially enter the channel of sales, and even the price or quantities may be anticipated. Such offer for sale activities could very well constitute patent infringement before the original brand-name drug's patent expires."
Therefore, the entire industry is in chaos and confusion: the me-too drug manufacturers have obtained NMPA approval by its own effort, and are qualified for marketing; while brand-name pharmaceuticals' original patents are still in force which may be infringed by the marketing of the me-too drugs. Any infringement damages after the me-too drugs are marketed, patent owners, me-too drug manufacturers and patients will have to foot the bill together, which is bad for the transformation of the entire pharmaceutical industry. The judiciary will also have to face tremendous social pressure for the public interest consideration.
Then, is early marketing of me-too drugs during the valid period of a patent indeed a disregard of potential patent infringement?
"By our observation, there has been no sign of large scale of marketing activities for me-too drugs. We speculate that they are also pretty cautious." Li Ming said, "by now, we are communicating with the authorities, and may take legal steps for enforcement later."
China IP learned that a few brand-name pharmaceuticals being followed on have started considering litigation. "Litigation, of course, shall be a conprehensive and strategic consideration. Besides the corporate strategies, it also will take into consideration of the brand-name drug in the corporate product position," says Li Ming. AstraZeneca has filed lawsuits this year against several me-too drug manufacturers that have obtained approval before patent expiration; Bayer has been actively responding to patent challenges on the one hand, and on the other carried out extensive work of collecting evidence for use in seeking injunctive relief once infringement activities take place.
Experts' proposal: judicial remedies and three-way joint efforts
Despite active response to series of lawsuits, Xu Feng indicated that litigation may not be the best choice. "It'll take more than a month to prepare for filing a lawsuit, and trial may be four months later. The cycle of a patent infringement lawsuit may easily last a long time. When a decision is made, the patent may have been long expired." Xu Feng said, "in the end, it is the insufficiency and inefficacy from lack of a rapid remedy. Even for intentional infringing enterprises, the effect of law is slow and overdue."
Some experts suggested that judicial authorities could render rewards of larger amount of damages to compensate for the loss. But Li Ming expressed that there was natural price difference between brand-name drugs and me-too drugs. It would not be realistic if damages be assessed on gross profit of brand-name drugs. "In China, damages are usually assessed by the statutory amount or based on illegal gains. But if it is based on the other side's profit, it will never cover the loss for brand-name pharmaceuticals," said Li Ming.
Timely and effective injunction is what brand-name pharmaceuticals would hope for. As is known to all, the courts keep a very high hurdle for preliminary and interlocutory injunctive relief, and the patentees have to clear many obstacles to obtain injunctions. "In the current state of affairs for domestic judicial remedies, a brand-name drug manufacturer can do nothing once a me-too drug is marketed despite the risks, and no injunctive relief is available," commented Liu Hongqiang. "In the pharmaceutical industry, the courts should give reasonable consideration to the uniqueness that distinguishes it from other industries. Once me-too drugs are put into market before the original patents expire, it will bring down the price for brand-name drugs and cause market erosion, which will definitely cause irreparable harm."
In fact, China IP found that the me-too drug manufacturers would not totally disregard potential patent infringement liabilities. "The key is lacking of relevant laws and mechanisms. We also hope for clear and executable laws and orderly market competition," one of the me-too drug manufacturer said.
On Aug 15, 2019, Beijing Intellectual Property Institute conducted an Expert Symposium on Patent Issues in Me-too Drug Approval and Marketing, during which experts offered views against status check by drug, healthcare and medicare authorities on patents. They suggested that in the absence of a patent linkage system in China, reliance on a "joint conference mechanism" could enhance information exchanging and sharing among departments. Where a brand-name drug's patent is not expired and its me-too drug is approved through technical examination, it may be considered as a practical measure to stamp on the approval document a date for starting production or sale, which shall be postdate the patent expiration date.
Finding a solution is the common goal of all parties.To break out this haze, without destruction there can be no construction.
Source: China IP Magazine, Issue 94









